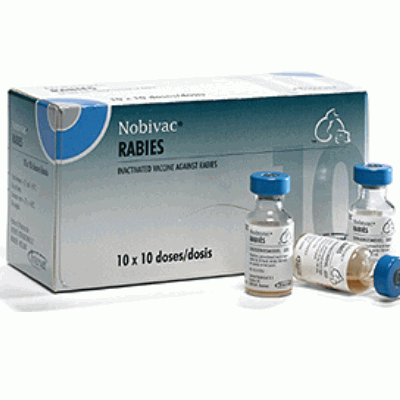
Nobivac Rabies
Nobivac Rabies is an inactivated vaccine against rabies for healthy dogs, cats, cattle, sheep, goats, horses, ferrets, foxes, and in principle all healthy mammals.The vaccine is highly immunogenic and offers complete protection that lasts at least 3 years.
Description
Nobivac Rabies
Brand Name: Nobivac Rabies
Manufactured by: MSD Animal Health
Dosage form: Injectable / Suspension for injection.
Presentation:
1 ml (1 Dose) x 10 Vials – Box
1 ml (1 Dose) x 50 Vials – Box
10 ml (10 Dose) x 5 Vials – Box
Ingredients & Composition:
Each 1 ml vial / Dose of Nobivac Rabies contains
Rabies Strain (Pasteur RIVM) with potency ≥ 2 IU of the inactivated rabies virus, adjuvanted with aluminiumphosfate.
Preservative: Thiomersal 0.01%
Description / Action:
Nobivac Rabies is an inactivated vaccine against rabies for healthy dogs, cats, cattle, sheep, goats, horses, ferrets, foxes, and in principle all healthy mammals. It is an adjuvanted, inactivated vaccine against rabies, prepared in cell culture. The seed virus of Nobivac Rabies is a highly immunogenic, fixed strain of rabies virus which is originated from Louis Pasteur’s original isolate in 1882. This inactivated virus obtained with repeated cell culture is formulated with a highly purified adjuvant and is packaged in liquid form. The vaccine is highly immunogenic and offers complete protection that lasts at least 3 years.
Class: Biologicals / Rabies Vaccine (Liquid Vaccine)
Indication / Uses:
Nobivac Rabies is indicated for
- Approved for active immunization of healthy dogs, cats, cattle, sheep, goats, horses, ferrets, foxes, and in principle all healthy mammals against rabies.
- It can be used for both prophylactic immunization & post-bite therapy.
Dosage and administration:
Vaccination Programme: Prophylaxis (Pre-exposure)
| RECOMMENDED VACCINATION PROGRAMME | Dogs/Cats | Cattle/Sheep /Horses | Ferrets |
| Primary vaccination at an age of more than | 3 months* | 6 months* | 3 months* |
| Dose | 1 ml | 2 ml | 1 ml |
| Revaccination every | 3 years** | 2 years** | 1 year** |
| Route of administration | s.c. or i.m. (dogs) | i.m. | s.c. |
* Primary vaccination can be administered at an earlier age, but then a repeat vaccination must be given at the age of 3 or 6 months depending on the species. A repeat dose after primary vaccination should be administered 1 year later.
** Recommended revaccination interval is based on challenge experiments’ results. Local regulations in force may require earlier revaccination in different countries.
** Annual revaccination is recommended in endemic areas.
Results from serological investigations indicate that vaccination of sheep, goats, and foxes provides protection for at least one year.
Vaccination Programme: Post-Bite treatment (Post-exposure Prophylaxis)
In all species, a repeated single dose of vaccine should be administrated (by the above-recommended route) according to the following schedule. (6th dose at day 90 if needed).
| Dose number | Timing |
| 1st | Day 0 (as soon as possible following exposure /bite) |
| 2nd | Day 3 |
| 3rd | Day 7 |
| 4th | Day 14 |
| 5th | Day 28 |
Duration of action: Studies have demonstrated that a single dose (1 ml) met federal guidelines for the protection of dogs and cats against virulent challenges administered 3 years after vaccination.
A single vaccination at 12 weeks of age will protect both cats and dogs for a period of at least 3 years.
If the first vaccine is given below the age of 12 weeks in healthy animals, there is possible interference with maternal antibodies, and for this reason, revaccination should be performed at the age of 12 weeks or older.
Immunity
After 3 weeks of vaccination in dogs, peak antibody titres have been demonstrated to occur. Studies conducted for the duration of immunity demonstrated that a 1 ml dose met federal guidelines for the protection of dogs, cats, and ferrets against virulent challenge administered more than a year after vaccination.
Overdoses / Side effects / Contraindications /Warnings:
After subcutaneous administration occasionally, a transient palpable nodule, vaccinoma or fever may occur at the site of injection. Such type of reactions can occur after the injection of any foreign protein and is, in most cases, self-limiting.
Generalized hypersensitivity or occasionally a mild, anaphylactic-type hypersensitivity reaction following administrations may occasionally occur. In this event administration of Adrenaline injection by subcutaneous route may be indicated and should be followed with appropriate supportive therapy. Vaccinate healthy animals only.
Always consult your vet if you see any adverse reaction. Contraindications -None, Special warnings – None. Do not vaccinate within 21 days before slaughter.
Pharmaceutical precautions / Instructions:
Store in a cool and dry place or refrigerator at 2-8°C. Prolonged exposure to higher temperatures may adversely affect potency. Do not freeze. Protect from light. Use sterile injection equipment. Shake before and during use. Vaccine should be allowed to reach ambient temperature (15-25°C) before use. 10-dose presentation should be used within one working day once it has been broached. For animal treatment only. No Special precautions to be taken by the person administering the veterinary medicinal product to animals.
In case of human exposure, contact a physician.
Nobivac Rabies suspension can be used to reconstitute the freeze-dried canine vaccines of the Nobivac series. It can be administered with the Nobivac leptospirosis vaccines at the same time but at a different administration site.
Safety and efficacy information of this vaccine is not available when used with any other veterinary medicinal product except the products mentioned above.
Do not mix with any other veterinary medicinal product except the vaccines of the Nobivac series mentioned above.
Safety: Age / Pregnancy/ Withdrawal:
Nobivac Rabies vaccine is produced on an established cell line so it has safety advantages over inactivated brain-origin rabies vaccines. Tissue-origin vaccines can lead to autoimmune disease because they contain extraneous protein in addition to rabies antigen.
A vaccine from established cell line has of consistent potency from serial to serial. Nobivac Rabies cell line has been extensively tested for freedom from contaminating agents.
Proven to be uniformly safe in experimental tests, with no significant adverse reactions in extensive clinical trials. Nobivac Rabies is safe in pregnant bitches. Withdrawal period not applicable.
Always follow recommended dose.
Habit forming: No, But Protocol should be followed according the guidelines.
Substitute: Rabisin, Canishot RV-F, Canvac – R, Nobivac Rabies, Canigen Rabies, Defensor, Raksharab
Note: In order to provide sufficient time to meet the requirements of the Pet Passport travel scheme it is recommended that vaccination against rabies should be done at least one month before you travel to ensure animals are immune before embarking.
Only logged in customers who have purchased this product may leave a review.



Reviews
There are no reviews yet.