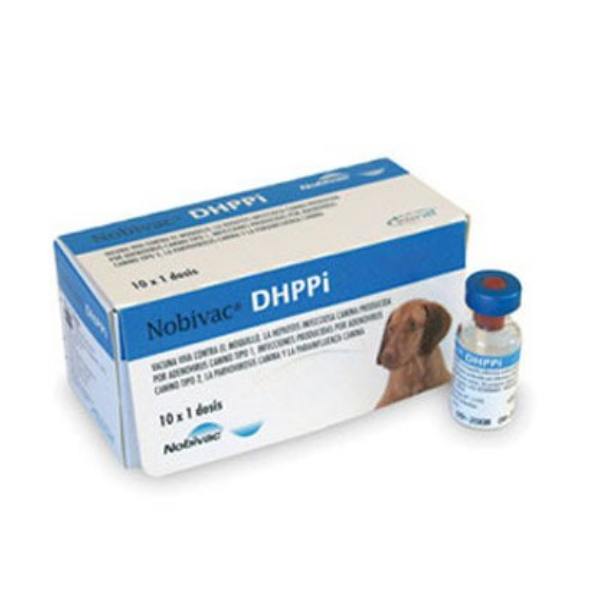
Nobivac DHPPi
Nobivac DHPPi is a 5 in 1 live attenuated freeze-dried vaccine for the immunization of dogs against canine distemper virus (CDV), canine adenovirus type 1 (CAV1), and 2 (CAV2), canine parvovirus (CPV), and canine parainfluenza virus (Cpi). The only vaccine with the 10 weeks final vaccination claim. Nobivac DHPPi is a core vaccine which contains attenuated antigens to stimulate active immunity in dogs.
Description
Nobivac DHPPi
Brand Name: Nobivac DHPPi
Manufactured by: MSD Animal Health (Intervet)
Dosage form: Injectable (Freeze dried Plug with Diluent)
Presentation:
1 ml (1 Dose) x 10 Vials – Box (Commonly available)
1 ml (1 Dose) x 50 Vials – Box
In combination with an equal number of vials with Nobivac Solvents, Nobivac L, Nobivac LR. Nobivac DHPPi, and Nobivac Solvent may be packed together with the vaccine or separately. In some countries pack of 5 or 25 vials also available.
Ingredients & Composition:
Each 1 ml vial / Dose of Nobivac DHPPi contains
Each dose contains live attenuated strains of:
Canine Parvo virus (strain C 154) ≥107 TCID50
Canine Distemper virus (strain Onderstepoort) ≥ 104 TCID50
Canine Adeno virus type 2 (strain Manhattan LPV3) ≥ 104 TCID50
Canine Para-influenza virus (strain Cornell) ≥105.5 TCID50
Description / Action:
Nobivac DHPPi is a live attenuated freeze dried vaccine for the immunization of dogs against canine distemper virus (CDV), canine contagious hepatitis / infectious canine hepatitis (ICH) caused by canine adenovirus type 1, canine parvovirus (CPV), and respiratory disease caused by canine parainfluenza (CPi) and canine adenovirus type 2 (CAV2).
The combination 5 in 1 vaccine against canine distemper virus (CDV), canine adenovirus type 1 (CAV1) and 2 (CAV2), canine parvovirus (CPV), and canine parainfluenza virus (Cpi). The only vaccine with the 10 weeks final vaccination claim.
Nobivac DHPPi is a core vaccine that contains attenuated antigens to stimulate active immunity in dogs.
Core Canine Vaccination
Vaccination is an integral part of a preventative health care programme. All dogs should be vaccinated against three core diseases i.e.– canine distemper, infectious hepatitis, and canine parvo virus. These diseases can have devastating consequences for a dog and it is easy to prevent them by regular vaccination.
The first “high titre, low passage” vaccine against parvovirus – Nobivac ParvoC – was launched in 1987 containing a patented strain of parvovirus called C154. The vaccine was the first parvo vaccine able to breakthrough levels of maternally derived antibodies. It was a revolution in canine vaccination.
Nobivac range still provides leading technology and premium parvo protection with vaccines that now provide proven protection against all known types of canine parvovirus (including types 2a, 2b, and 2c). It provides protection in puppies from as early as 10 weeks of age, and the convenience of single vaccination in dogs with unknown vaccination histories
Class: Biologicals / DHPPi Vaccine (Liquid Vaccine)
Indication / Uses:
Nobivac DHPPi is indicated for Active immunization of healthy dogs against
• To prevent mortality and clinical signs caused by Canine distemper virus (CDV) infection.
• To prevent mortality, clinical signs, and viral excretion following canine parvovirus infection (CPV).
• To reduce clinical signs and viral excretion caused by canine parainfluenza virus (CPi).
• To reduce clinical signs of respiratory disease and viral excretion following canine adenovirus type 2 (CAV2).
• Entities such as respiratory tract infections.
• It protects and reduces clinical signs of infectious hepatitis and viral excretion due to canine adenovirus type 1 infection.
Dosage and administration:
Reconstitute the contents of one vial of Nobivac DHPPi in one vial of Nobivac solvent or Nobivac Lepto or Nobivac Rabies or Nobivac RL. The reconstituted vaccine should be used within 30 minutes. Reconstituted vaccine should be injected subcutaneously.
The basic vaccination schedule of puppies can be completed with Nobivac DHPPi, and it is indicated for annual or bi-annual revaccination as well.
Avoid contamination of vaccine with traces of chemical sterilizing agents. Disinfectant or spirit should not be used to disinfect the skin prior to inoculation.
Vaccination Programme:
Primary course vaccination
In dogs of 10 weeks of age or older A single injection should establish active immunity to canine distemper, infectious canine hepatitis, and disease caused by canine parvovirus infection.
A first dose may be given to puppies from 6 weeks of age where earlier protection is required. The final dose at 10 weeks of age or older is generally recommended because maternally derived passive antibodies can interfere with the response to vaccination in case of early vaccination at 6 weeks of age. Animals should be vaccinated twice for optimal response to the parainfluenza component at 2-4 weeks apart with the final vaccination at 10 weeks of age or more.
Booster vaccination
It is recommended that dogs be revaccinated every 3 years with canine distemper virus, canine adenovirus and canine parvovirus and every year against canine parainfluenzavirus.
To complete the basic vaccination schedule in puppies, and for annual or biannual revaccination Nobivac DHPPi is indicated.
Each of the Nobivac vaccines fits into a larger overall vaccination programme. The individual’s age, health (including maternal immunity), and local environmental conditions should be considered in the formulation of a vaccination programme.
Suggested Vaccination Protocol
1. Vaccination commencing at 6- 8 weeks of age. (Two doses)
Age Vaccine
6 – 8 weeks Nobivac DHPPi
10 weeks Nobivac DHPPi
*Core vaccination for dogs, CPV, CDV and CAV.
**Puppies must be of 10 weeks of the minimum age at final vaccination.
2. Vaccination commencing at 10 weeks of age or older. (One dose)
Age Vaccine
10 weeks or older Nobivac DHPPi
A single dose from the age of 12 weeks is sufficient to establish immunity for the parainfluenza component of Nobivac DHPPi,
In animals younger than 12 weeks, 2 doses of this component are recommended so that the first dose is administered from the age of 8 weeks onwards, and the second dose, 2 – 4 weeks later.
In summary:
Animals 6 – 10 weeks of age: 2 doses of Nobivac DHPPi, 2 – 4 weeks apart such that the second dose is given from 10 weeks of age.
For protection against parainfluenza, the first dose should be administered = 8 weeks.
Animals of 10 – 12 weeks of age: 2 doses of Nobivac DHPPi, 2 – 4 weeks apart.
Animals of 12 weeks of age: 1 dose of Nobivac DHPPi.
Revaccination
It is recommended that a single booster dose is given as follows:
Canine distemper, adenovirus and parvovirus: every 3 years.
Canine parainfluenza: every year.
Duration of immunity:
The components of Nobivac DHPPi are shown to guard against CDV, CPV and CAV for at least 3 years and canine parainfluenza virus with 1 year duration of immunity.
However, the manufacturer recommends that the 1st year annual booster should be a ‘full’ DHPPi and Lepto vaccination.
The final vaccination of puppies can be carried out at an age of 10-12 weeks with Nobivac DHPPi, allowing for early socialization and prevention of behavioural problems.
Onset of immunity:
An onset of immunity to the canine distemper virus, canine adenovirus, and canine parvovirus vaccine components of 1 week and onset of immunity of 4 weeks to the canine parainfluenza virus vaccine component has been demonstrated after the administration of the vaccine.
Overdoses / Side effects / Contraindications /Warnings:
Only healthy dogs should be vaccinated. Dogs incubating the disease at the time of vaccination may result into failure or inefficacy of the vaccine.
Some dogs may be immunologically incompetent and fail to respond to vaccination. Animals those have been given or treated with the corresponding anti-serum or immunosuppressive drugs should not be vaccinated until an interval of at least 4 weeks has elapsed.
At the site of injection a diffuse swelling, up to 5 mm in diameter, maybe observed after administration. This swelling may be hard and painful and lasts for up to 3 days post-injection occasionally.
In the case of hypersensitivity reactions administer an antihistamine, corticosteroid, or adrenaline, without delay and by the most immediate route; occurring following vaccination though it is rare.
A response similar to that following a single dose may be seen after administration of an overdose. In some dogs the swelling may be observed for a longer period or more painful.
After inoculation or administration, canine parvovirus vaccine strain may be shed at very low levels for up to 8 days. No need to separate unvaccinated dogs from contact with recently vaccinated individuals because there is no evidence of any reversion to virulence of the vaccine strain.
Nobivac Rabies and Lepto liquid vaccines can be used to reconstitute the freeze-dried Nobivac DHPPi. No information is available on the use of this vaccine with any other, except these two vaccines. Other immunological product administration is not recommended within 14 days before or after vaccination with Nobivac DHPPi.
Maternal antibody interference can reduce the efficacy of the CDV, CAV2, CPV, and CPi components of the vaccine. In the presence of maternal antibody levels to CDV, CAV2, CPV, and CPi that are likely to be encountered under field conditions, the vaccine has been proved to be of benefit against virulent challenge.
Pharmaceutical precautions / Instructions:
Store in a cool and dry place or refrigerator at 2-8°C. Care should be taken to avoid repetitive exposure to high ambient temperatures or prolonged exposure to high temperatures following withdrawal from the refrigerator prior to use. Vaccine potency in hot summer conditions can be severely reduced within a few hours. Do not freeze. Protect from light. Use sterile injection equipment. Shake before and during use. After reconstitution: use within 30 minutes. For animal treatment only. No Special precautions to be taken by the person administering the veterinary medicinal product to animals.
In the case of human exposure, contact a physician.
Waste material should be disposed of by boiling, incineration or immersion in an appropriate disinfectant approved for use by the competent authorities.
Safety: Age / Pregnancy/ Withdrawal:
In adult dogs, parainfluenza challenge was not able to produce clinical signs of kennel cough. An anamnestic response was seen in dogs given a booster one year after primary vaccination but the duration of immunity could not, therefore, be demonstrated.
Prior to exposure to high-risk environments such as kennelling, showing, or mixing with dogs of unknown vaccination history Revaccination against parainfluenza is recommended.
Experience has shown that the maternal antibody status of pups within a litter varies greatly and reliance should not be placed on the serological examination of the bitch alone.
A good immune response is reliant on the reaction of an immunogenic agent and a fully competent immune system. Poor storage or inappropriate administration will reduce the immunogenicity of the vaccine antigen. A variety of factors including poor health, nutritional status, genetic factors, concurrent drug therapy, and stress may compromise the Immunocompetence of the animal.
Nobivac DHPPi vaccine can be used in pregnant bitches which have previously been vaccinated with the CDV (strain Onderstepoort), CAV2 (strain Manhattan LPV3), CPV (strain 154) and CPi (strain Cornell) antigens included in the Nobivac vaccine series.
Withdrawal period is not applicable.
Always follow the recommended dose.
Habit forming: No, But Protocol should be followed according to the guidelines.
Substitute: Canigen DHPPi
Only logged in customers who have purchased this product may leave a review.



Reviews
There are no reviews yet.