Apoquel Tablets
Apoquel Tablets contain oclacitinib which is a Janus kinase (JAK) inhibitor. It can prevent many cytokines from functioning that are dependent on the activity of the JAK enzyme. The target cytokines for oclacitinib are those that contribute to allergic reactions or pruritus or that are pro-inflammatory. However, oclacitinib may also exert effects on other cytokines (for example, those involved in host defence or haematopoiesis) with the potential for unwanted effects.
Description
Apoquel Tablets
Oclacitinib Maleate Tablets for Dogs
Manufactured/ Marketed by: Zoetis
Country of Origin: USA
Doses form: Tablets
Presentation:
Bottles or Blister packs of 20, 50 or 100 tablets per pack.
Not all pack sizes may be available for marketing.
Ingredients & Composition:
Each film coated tablet contains
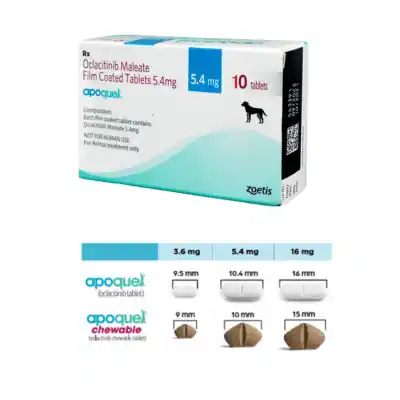
Oclacitinib Maleate 3.6 mg, 5.4 mg or 16 mg.
Film-coated tablets, oblong in shape and white to off-white in colour, bearing the letters “AQ,” “M,” or “L” on both sides and a score line on the other. The letters “M” and “L” refer to the different strengths of tablets: “M” on the 5.4 mg tablets, and L on the 16 mg tablets. You can split the tablets into two equal parts.
Description:
Apoquel Tablets contain oclacitinib which is a Janus kinase (JAK) inhibitor. It can prevent many cytokines from functioning that are dependent on the activity of the JAK enzyme. The target cytokines for oclacitinib are those that contribute to allergic reactions or pruritus or that are pro-inflammatory. However, oclacitinib may also exert effects on other cytokines (for example, those involved in host defence or haematopoiesis) with the potential for unwanted effects.
Class: Immunosuppressant
Indication / Uses:
For treatment of pruritus associated with allergic dermatitis in dogs.
For treatment of clinical manifestations of atopic dermatitis in dogs.
Dosage and Administration:
Apoquel dose for Initial therapy:
0.4 to 0.6 mg /kg bodyweight, administered orally, twice daily for up to 14 days.
Apoquel dose for maintenance therapy (after the initial 14 days):
0.4 to 0.6 mg /kg bodyweight should then be given only once a day.
The responsible veterinarian should perform an individual benefit-risk assessment before determining if long-term maintenance therapy is necessary.
These tablets can be given with or without food. Please see dosing table below for the number of tablets required to achieve the recommended dose. The tablets are breakable along the score line.
Apoquel Dosing Chart for Dogs
Bodyweight (kg) Strength and number of tablets:
3.6 mg 5.4 mg 16 mg
3.0-4.4 ½
4.5-5.9 ½
6.0-8.9 1
9.0-13.4 1
13.5-19.9 ½
20.0-26.91 2
27.0-39.9 1
40.0-54.9 1 ½
55.0-80.0 2
Dogs should be carefully observed following administration to ensure that each tablet is swallowed.
Target species:
For Oral Use in Dogs Only
Contraindications:
Do not use in cases of hypersensitivity to oclacitinib or to any of the excipients.
Do not use in dogs less than 12 months of age or less than 3 kg bodyweight.
Do not use in dogs with evidence of immune suppression such as hyperadrenocorticism or with evidence of progressive malignant neoplasia as the active substance has not been evaluated in these cases.
Adverse reactions:
The common adverse reactions seen up to day 16 of the field trials are listed in the following table:
Adverse reactions like Diarrhoea, Vomiting, Lack or loss of appetite (Anorexia), New cutaneous or subcutaneous lumps, Weakness (Lethargy), Excessive thirst (Polydipsia) were observed in atopic dermatitis study up to day 16 and pruritus study up to day 7, respectively.
After day 16, the following adverse reactions have been observed: pyoderma and non-specified dermal lumps have been observed very commonly; otitis, vomiting, diarrhoea, histiocytoma, cystitis, yeast skin infections, pododermatitis, lipoma, polydipsia, lymphadenopathy, nausea, increased appetite and aggression have been observed commonly.
Treatment-related clinical pathology changes were restricted to an increase in mean serum cholesterol and a decrease in mean leukocyte count, however, all mean values remained within the laboratory reference range. The decrease in mean leukocyte count observed in oclacitinib-treated dogs was not progressive, and affected all white blood cell counts (neutrophil, eosinophil and monocyte counts) except lymphocyte counts. Neither of these clinical pathology changes appeared clinically significant.
The development of papillomas was noted in a number of dogs in a laboratory study. Anaemia and lymphoma have been reported very rarely in spontaneous reports.
The frequency of adverse reactions is defined using the following convention:
very common (more than 1 in 10 animals treated displaying adverse reaction(s))
common (more than 1 but less than 10 animals in 100 animals treated)
uncommon (more than 1 but less than 10 animals in 1,000 animals treated)
rare (more than 1 but less than 10 animals in 10,000 animals treated)
very rare (less than 1 animal in 10,000 animals treated, including isolated reports).
If you notice any side effects, even those not already listed in this package leaflet or you think that the medicine has not worked, please inform your veterinary surgeon.
Overdose and side effects of apoquel (symptoms, emergency procedures, antidotes):
Oclacitinib tablets were administered to healthy, one year old Beagle dogs twice daily for 6 weeks, followed by once per day for 20 weeks, at 0.6 mg/kg bw, 1.8 mg/kg bw and 3.0 mg/kg bw for a total of 26 weeks. Clinical observations that were considered likely to be related to oclacitinib treatment included: alopecia (local), papilloma, dermatitis, erythema, abrasions and scabbing/crusts, interdigital “cysts”, and oedema of the feet. Dermatitis lesions were mostly secondary to the development of interdigital furunculosis on one or more feet during the study with the number and frequency of observations increasing with increasing dose. Lymphadenopathy of peripheral nodes was noted in all groups, increasing in frequency with increasing dose, and was frequently associated with interdigital furunculosis.
Papilloma was considered treatment related, but not dose related.
There is no specific antidote and in case of signs of overdose the dog should be treated symptomatically.
Special warning(s):
Special precautions for use in animals:
Oclacitinib modulates the immune system and may increase susceptibility to infection and exacerbate neoplastic conditions. Dogs receiving Apoquel tablets should therefore be monitored for the development of infections and neoplasia.
When treating pruritus associated with allergic dermatitis with oclacitinib, investigate and treat any underlying causes (e.g. flea allergic dermatitis, contact dermatitis, food hypersensitivity). Furthermore, in cases of allergic dermatitis and atopic dermatitis, it is recommended to investigate and treat complicating factors, such as bacterial, fungal or parasitic infections/infestations (eg flea and mange).
Given the potential for effects on certain clinicopathological parameters, periodic monitoring with complete blood counts and serum Biochemistry is recommended when dogs are on long-term treatment.
Special precautions to be taken by the person administering the veterinary medicinal product to animals:
Wash hands after administration.
In case of accidental ingestion, seek medical advice immediately and show the package leaflet or label to the physician.
Interaction with other medicines:
No drug interactions were observed in field studies where oclacitinib was administered concomitantly with veterinary medicinal products such as endo and ectoparasiticides antimicrobials and anti-inflammatories.
The impact of oclacitinib administration on vaccination with modified live vaccines, canine, parvovirus (CPV), canine distemper virus (CDV) and canine parainfluenza (CPI) and inactivated rabies vaccine (RV), on 16-week-old vaccine naive puppies has been studied. An adequate immune response (serology) to CDV and CPV vaccination was achieved when puppies were administered oclacitinib at 1.8 mg/kg bodyweight (bw) twice daily for 84 days. However, the findings of this study indicated a reduction in serological response to vaccination with CPI and RV in puppies being treated with oclacitinib compared to untreated controls. The clinical relevance of these observed effects for animals vaccinated while being administered oclacitinib (in accordance with the recommended dosing regimen) is unclear.
Safety: Age / Pregnancy:
The safety of the veterinary medicinal product has not been established, during pregnancy and lactation, or in breeding male dogs, therefore its use is not recommended during pregnancy, lactation or in dogs intended for breeding.
Withdrawal period(s):
Not applicable.
Pharmaceutical precautions / Instructions:
Keep out of the sight and reach of children. Not for human use. For Animal treatment only.
Store below 30 °C. Any remaining half tablet should be placed back in the opened blister and stored in the original cardboard carton, or in the HDPE bottle (for a maximum of 3 days). Do not use this veterinary medicinal product after the expiry date which is stated on the blister or bottle after EXP.
Special precautions for the disposal of unused product or waste materials, if any:
Medicines should not be disposed of via wastewater or household waste.
Ask your veterinary surgeon how to dispose of medicines no longer required. These measures should help to protect the environment.
Warning:
To be sold by retail on the prescription of a “Veterinary Doctor” only
Substitute: Updating Soon
Only logged in customers who have purchased this product may leave a review.




Reviews
There are no reviews yet.